Proctalgia in Pudendal Nerve Entrapment Syndrome: A Pathology to Know
José Felipe Reoyo Pascual, Rosa M Martínez Castro, Raquel León Miranda, Carlos Cartón Hernández and Xandra Valero Cerrato
DOI10.21767/2471-299X.1000044
José Felipe Reoyo Pascual*, Rosa M Martínez Castro, Raquel León Miranda, Carlos Cartón Hernández and Xandra Valero Cerrato
Service of General Surgery and Digestive, Hospital University of Burgos, Burgos, Spain
- *Corresponding Author:
- José Felipe Reoyo Pascual
Service of General Surgery and Digestive
Hospital University of Burgos, Av. Islas Baleares s/n., Burgos, Spain
Tel: +34-647246428
E-mail: jreoyo@saludcastillayleon.es
Received Date: January 24, 2017; Accepted Date: March 07, 2017; Published Date: March 14, 2017
Citation: Pascual JFR, Castro RMM, Miranda RL, et al. Proctalgia in Pudendal Nerve Entrapment Syndrome: A Pathology to Know. Med Clin Rev. 2017, 3:1. doi: 10.21767/2471-299X.1000044
Copyright: © 2017 Pascual JFR, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Pudendal nerve entrapment syndrome is a little known and under-diagnosed condition that includes urinary, sexual and proctological signs and symptoms. Its diagnosis is complex and requires sequential treatment. Proctalgia is a common symptom in the General Surgery field, and it can be attributed to pudendal nerve entrapment syndrome in a significant percentage of patients. Objective: The aim of our study is to determine the association between proctalgia and pudendal nerve entrapment syndrome, to check the immediate response to treatment by infiltration with a corticosteroidanesthetic injection, and to assess the results after 6 months follow-up. Design: An observational study was conducted to assess 53 patients affected by proctalgia in the context of a pudendal nerve entrapment syndrome, who were subjected to treatment with hydrodistentioncorticosteroid- anesthetic infiltration of the pudendal nerve and its terminal branches. Their immediate response, in clinical terms of pain, and after six months of follow-up was assessed. Results: From the data obtained in our study, is was observed that treatment with these injections showed an improvement in 79.25% of the patients in the period immediately after the injection, and 39.62% of patients maintained that improvement after six months. Conclusion: We conclude that, in the presence of proctalgia, the existence of a pudenda nerve entrapment syndrome has to be ruled out, and that treatment with corticosteroidanesthetic infiltration is an effective option that achieves an improvement in a significant percentage of patients.
Keywords
Pudendal nerve; Pudendal nerve entrapment; Pudendal neuropathy; Proctalgia
Introduction
Pudendal Nerve Entrapment Syndrome (PNES) is a condition that was described by Amarenco et al. [1]. Since then, many attempts have been made to determine its etiology, diagnosis, and treatment.
The pudendal nerve is complex, with three terminal branches: The inferior rectal nerve, perineal nerve, and the dorsal nerve of the penis/clitoris [2-4]. There are critical points of nerve entrapment: The sacrospinous and sacrotuberous ligament, the falciform process, and Alcock’s canal.
Causes associated with this entrapment are: professional/ sports activities that involve the sitting position [5,6], perineal injuries [7], gynecological causes [8-10], pelvis radiotherapy, rectal prolapse [11-13].
The main clinical symptom is unilateral or bilateral pain in nerve distribution region that gets worse throughout the day and on sitting down, and improves on lying down.
It can be associated with proctological (constipation, rectal foreign body), urinary (tenesmus, incontinence), or sexual symptoms (dyspareunia, painful ejaculation) [14-16].
Two signs are highlighted in the examination, the Tinel sign (pain triggered by compressing the nerve against the ischiatic spine), and the Rolling Test (rolling a fold of skin from the anus to the pubis in order to reproduce the pain) [17].
There are neurophysiological tests, such as the distal motor latency test (St. Mark´s) that evaluate the motor function of the nerve [18-20]. There are also well-established criteria, known as the Nantes criteria (Table 1) [21-23].
| NANTES CRITERIA |
|---|
| 1) Pain in the anatomical region of the pudendal nerve |
| 2) Gets worse on sitting down |
| 3) The patient is not awakened at night by the pain |
| 4) There is no subjective loss of sensitivity in the clinical examination |
| 5) There is a positive response to pudendal nerve block |
Table 1: Nantes criteria for the diagnosis of PNES.
The differential diagnosis, supported by imaging tests, is established between endometriosis, tumors, metastasis, and other lesions that cause compression of the Alcock’s canal [23,24].
Treatment initially includes symptomatic management of the pain with amitriptyline, gabapentin, pregabalin [25-27]. Another therapeutic mainstay is corticosteroidanesthetic injections, as will be discussed in detail later on [28-30]. Pelvic physiotherapy [31], botulin toxin etc. [32], have also been used. As regards surgical treatment, the transgluteal, transperineal, or transvaginal technique should be mentioned [33-36], which have the common objective of freeing the nerve from its entrapment.
PNES is a multidisciplinary research field that opens horizons in its diagnosis and treatment.
The use of stem cells is being studied, as well as pulsed radiotherapy, laparoscopy, etc., with the results fairly inconclusive [37].
Material and Methods
A prospective observational study has been conducted on 53 patients with proctalgia, who met the Nantes Criteria [33], thus being diagnosed with PNES (Table 1). An injection was given in the pudendal nerve according to a technique described below. The period of the study was from 1 June 2011 to 30 June 2013.
This study has three aims:
• To establish the relationship between proctalgia and PNES.
• To determine the immediate response to the treatment using anesthetic infiltration.
• To assess the post-infiltration clinical results and the efficacy of the treatment in a mediumterm follow-up (6 months).
In order to evaluate the results, Visual Analog Scale (VAS) scores at three different times were taken from the clinical notes:
• Baseline pain level.
• Immediate post-injection period.
• Evaluation at 6 months.
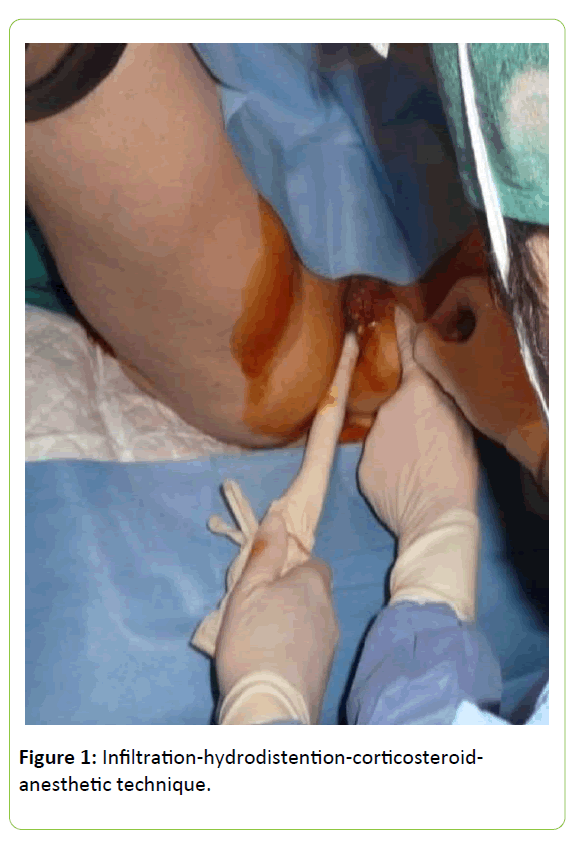
The technique used was infiltration-hydrodistention with corticosteroids and 0.2% ropivacaine at trunk level and at the three terminal branches of the nerve (Figure 1). The injection can be uni-or bilateral, as in our sample. The transdermal route in lithotomy is the anatomical position. Transanal ultrasound is used as a guide and neurostimulator of the peripheral nerves. The injection is repeated at one week after the first infiltration and two weeks after the second injection up to a total of 3 infiltrations. In that patient in whom no significant improvement is observed, a second round of infiltration is planned.
The patients were divided into two groups:
Non-responders: Those patients whose reduction of the score on the VAS was LESS THAN 30% at 6 months after the first infiltration.
Responders: Those patients whose reduction of the score on the VAS was GREATER THAN OR EQUAL TO 30% at 6 months after the first infiltration.
Results
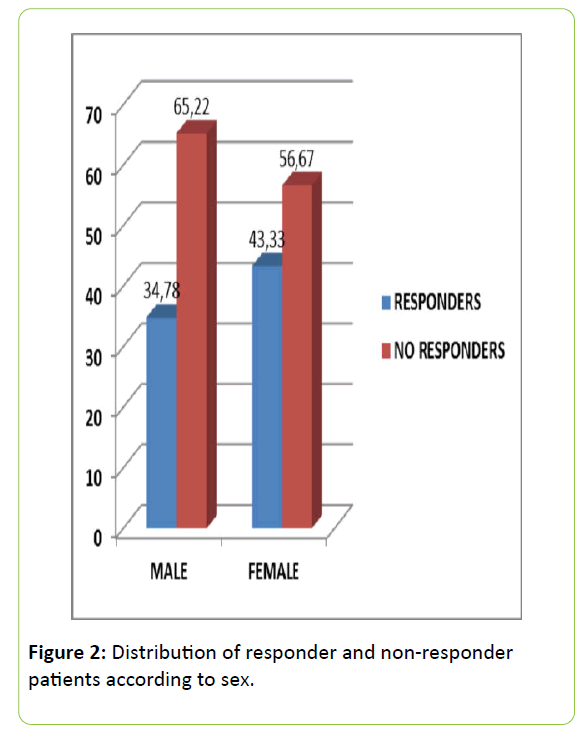
Of the 53 patients studied, 43.39% were male and 56.61% were female. The responders consisted of 34.78% males and 43.33% females (Figure 2). As regard the nerve branches affected (according to the symptomatology presented, sexual-anterior branch, urological middle branch, proctological-posterior branch), 64.15% had 3-branch involvement, 2-branch in 30.18%, and only posterior branch involvement in 5.67% of the cases. In the group with 3 branches involved, it was observed that there were a higher percentage of nonresponders compared to those with 2-branch involvement. The mean age of the patients studied was 56.8 years (24-85 years). As regards personal history, 21.74% of the male group had a history of anorectal surgery, and 34.78% had no history of interest, and in the women, added to these two large groups, was gynecological history (33.33%). The Tinel sign was positive in 98.1%, and only 37.73% of cases were positive in the Rolling Test, with no differences between responders and non-responders. The distal motor latency test of the nerve showed abnormal in 88.46% of cases, and it was observed that there were almost twice as many patients (19.05%) in the responders group with a normal test.
The mean pre-infiltration score on the VAS was 7.74 (a mean of 7.08 in the responders group, and 7.83 in the non-responders). The mean VAS score immediately after the infiltration was 1.19, with 79.25% of the patients having a score of 0. The mean score was 0.4 in the responders and 1.72 in the non-responders.
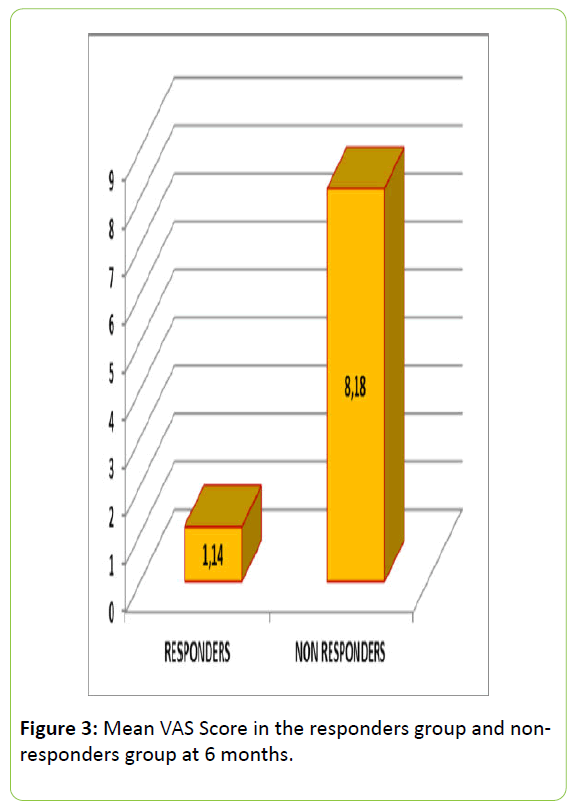
At 6 months it was observed that 32 (60.38%) patients were non-responders and 21 (39.2%) were responders. A pain score of 0 in the VAS was noted in 24.5% of the patients. The overall mean VAS score in this period was 5.39, a mean of 1.14 being observed in the responders group, and 8.18 in the non-responders group (Figure 3).
In the 159 infiltrations performed, only 4 (2.51%) complications were recorded: 2 amenorrhea, 1 vaginal bleeding, and one vasovagal syncope. Currently, 16 patients are receiving a second round of infiltrations.
No significant differences as regards response to treatment were observed on applying the variables of sex, history and affected branch, clinical signs, and St. Mark’s test to the statistics (Table 2). On the other hand, on comparing the mean pre-infiltration score on the VAS with the post-infiltration score at 6 months, a statistically significant difference was observed in the overall sample, as well as distinguishing between sexes, using the Wilcoxon test (p<0.00003).
| Variable | Test | P- Value | Signification? |
|---|---|---|---|
| Sex | Chi- Square | 0.337 | NO |
| Personnel History | Chi- Square | 0.336 | NO |
| Branches | V Cramer | 0.467 | NO |
| Clinical Signs | V Cramer | 0.432 | NO |
| ST. Marks | V Cramer | 0.467 | NO |
| VAS Pre/Post infiltration | Wilcoxon | 0.00003 | YES |
Table 2: Statistical study.
Discussion
PNES is a condition that was described for the first time in 1987 by Dr. Amarenco, giving it the name of Cyclist Perineal Palsy Syndrome [1]. Since then, Shafik, Bautrant, Robert, Beco, Benson… have shed light on various points associated with the syndrome, although there are still many aspects that need clarifying [2].
The series published have indicated that the incidence and prevalence is unknown, probably due to the under diagnosis of this condition, with the mean time to diagnosis being 4 years (range 1 to 15 years). During this time, patients are assessed by various specialists (surgeons, gynecologists, urologists, etc.), who, often due to lack of knowledge of this syndrome, do not give an accurate diagnosis, offering solutions for infections, etc. This prolongs the diagnosis time, with the resulting worsening of the quality of life of the patients and unnecessary waste of poorly directed health resources.
As regards the distribution by sexes, Lema and Ricci [38] indicated that PNES is present more frequently in females, stating that in the next few years in Europe and America, 1 in every 7 women will probably develop PNES. This is probably due, to a great extent, to etiological factors common to the female sex, such as the long list of gynecological causes [39]. The results of the present study agree with those already published, since 56.6% of our patients are women. It is also observed that 34.78% of the male group responds to the infiltrations, while 43.33% of the female group responds. From this it can be deduced that women respond better to the anesthetic infiltrations, although not reaching statistical significance.
The series already published show that there is no predisposition for PNES by any age group, an aspect confirmed in our sample [40].
As regards etiology, professional or sports activities that involve a long time in the sitting position are implicated, such as horse riding, cycling, etc. [1,6]. In our sample there are 4 professional cyclists, which make up 7.5% of the cases. There are gynecological-obstetrics causes such as, vaginal partum, episiotomy, pelvic and vaginal surgery [8-10], in which a higher incidence of PNES has been observed. In our series there were 33.3% of women with a gynecological history. As regards anorectal surgery, few studies have analyzed its relationship with PNES, although Bautrant et al. [41] indicated pelvic surgery as one of the most frequent causes of the syndrome. In our series, 18.86% had a history of anorectal surgery.
Other processes associated with the syndrome are perineal injuries [7], pelvic radiotherapy, rectal prolapse, etc., although none of these were present in our sample.
There are many cases in which no particular cause has been identified. Thus, in our sample, 33.33% of the women and 34.78% of the men had no history of interest. Proctalgia is the main constant in our study, to which might be added symptoms from other nerve branches [14-17]. Beco et al. [18], analyzed the relationship between fecal and urinary incontinence and perineal pain, observing that 71% of the patients presented with two of these symptoms, while 13% presented with the three of them. On the other hand, in our sample, 64.15% of the cases had 3-branch symptoms, 30.18% with 2-branch involvement, and 5.67% with only posterior branch involvement. It was observed that there was a higher percentage of non-responders (65.71%) in the group of patients that had 3-branch symptoms, compared to those with 2-branch involvement (50%), thus observing a poorer response in those patients with 3-branch involvement.
We consider the Tinel sign to be very valuable and very sensitive, since 98.1% of our patients were positive, with only one patient, a non-responder, showing negative for this sign, 9 therefore, also showing to be a specific sign. As for the Rolling Test [17], it was only positive in 37.73% of our sample, with no significant differences being observed between responders andnon-responders; therefore we consider that this sign is not very sensitive or specific.
The most important test in the neurophysiological study is the Distal Motor Latency Test of the nerve (St. Mark´s), which is based on measuring the nerve motor conduction velocity, and is not very useful for assessing the extent of the lesion [19,20]. The result was abnormal in 88.45% of the cases in our sample. If we distinguish between responders and non-responders, abnormal results were observed in 80.95% and 90.625%, respectively, confirming that this is a sensitive and specific test. On looking at our data, we deduce that the patients respond better when they have a normal St Mark´s test.
Imaging tests are fundamental for the diagnosis of exclusion. In our sample, Computed Tomography (CT) scan was performed in 14 cases, a Magnetic Resonance (MR) scan in all cases, and endorectal ultrasound in 49 patients. Mollo [21] used color Doppler of the internal pudendal artery to support the diagnosis.
The treatment of PNES is based on 3 mainstays; symptomatic treatment, corticosteroidanesthetic infiltrations, and surgical intervention.
All the patients in our series had received medical treatment based on amitriptyline and antiepileptics, such as pregabalin or gabapentin, which are widely used in the literature [26,27].
The basis of our study was to analyze the treatment with corticosteroid-anesthetic infiltrations [28-30,40,42]. It is a safe technique, since we have only observed 4 minor complications. As regards surgical treatment, this is not routine in Spain, although the Hospital de Vigo is starting to use a nerve decompression technique by a transgluteal approach.
We have used the VAS applied to proctalgia, as we consider that it is an easy method and faithfully reflects the reality. Using this, we obtained a mean pre-infiltration score of 7.74. In the responders, the mean VAS score was 7.08, and in the non-responders it was 7.83; thus in the pre-infiltration period we can already observe a slight tendency in the non-responders group to having a higher score.
The mean VAS score in the immediate period after the infiltration was 1.19, and it has to be mentioned that in 42 of the 53 patients studied had recorded a score of 0, from which it can be deduced that the technique is effective in the short term. However, on analyzing the progress, it is observed that many patients who had shown a favorable response, with the passing of time, the symptoms returned and achieved a score equal to or higher than the pre-infiltration scores. This leads us to believe that the immediate post-infiltration VAS score is not a good indicator of the long-term response.
At 6 months after the first infiltration, a mean VAS score of 5.39 was obtained. In 13 patients (24.5% of the cases) the score had been 0. There were 32 non-responder patients (60.38%) and 21 responder patient (39.62%). It may be deduced from these figures, although the response may not be complete in the long-term, the responders maintain a reasonable score after the first infiltration. This fact is of considerable significance, since many of our patients, have had disabling symptoms for years, which were refractory to all types of conventional treatments, forming part of a long journey to specialists unable to offer a solution to their problems. Therefore, an improvement of more than 30% in their level of pain on the preinfiltration VAS, assumes an alleviation, satisfaction, and gratitude that we consider a great advance and a solution, which although sometimes may not be total, significantly improves the quality of life.
On applying the Wilcoxon test and comparing the pre-infiltration VAS scores with the postinfiltration scores obtained at 6 months, it can be observed that the VAS values have decreased significantly, which confirms that the hydrodistention-corticosteroid-anesthetic infiltration is a technique that offers complete improvement, and a partial one in other cases, to a significant percentage of patients.
There are few references in the literature that evaluate a follow-up longer than the immediate period after the first infiltration, as in our study. Thus, Ricci et al. [40] analyzed CT-guided pudendal nerve infiltration in series of 5 patients, stating that the pain decreased in all of them after the infiltration, but adding that only the follow-up could provide evidence of maintaining good results. Similar findings were obtained by Mc Donald and Spigos [43], but there were no long-term data. Those of Fannucci et al. [44] agree with our results, in that infiltration is a safe treatment, obtaining a clinical effectiveness higher than ours (92% at 12 months).
Dehkharghani et al. [45] demonstrated a high success rate, but were based on the immediate postinfiltration results, with no long-term data. Filler [46] opted to use MR to obtain accurate images of the nerve and to better direct the infiltration. As mentioned previously, our technique was ultrasound-guided. Rofael et al. [34] showed that ultrasound allowed anatomical points to be visualized, as well as observing the spreading of the local anesthetic and improving the accuracy of the technique, and reducing complications to the minimum.
Choi et al. [47] used fluoroscopy to improve the viewing, referring to a success rate of 100%, based on the reduction of 2 points on the VAS measured at 12 months after infiltration. In our opinion, it should be pointed out that we have not considered a reduction of 2 points on the VAS as a success, since we have based it on a percentage reduction from the baseline VAS score, considering this to be a better reflection of the reality. Le Tallec de Certains [48] analyzed the progression of the pain and the response to the infiltration over time, observing better results in cases of neuralgia, with the pain starting to lower at 1 year. On analyzing these data, it could be that the short-term “acute” pain may be due to resolvable, transient, causes, whereas “chronic” pain could be due to “permanent”, difficult to resolve, causes.
There are currently two lines to follow after the failure of the first infiltration. These are, a new round of injections or to contemplate decompression surgery.
In our case, we opted for a second round of injections, on being a less aggressive and conservative treatment. Popeney et al. [49] chose decompression surgery, demonstrating good results in 50 patients. Robert et al. [50] showed the better results with surgical vs. medical treatment. New lines of research, such as the use of pulsed radiofrequency or sacral neuromodulation are being investigated in some specialized pain units.
Conclusion
In conclusion, PNES is a painful and invalidating condition of unknown incidence and prevalence. It is underdiagnosed and is more common in females, and can appear at any age. There is a high percentage of patients with a gynecological history, anorectal surgery, and of no particular interest. According to our data, 3-branch involvement is the most frequent and is the group that responds worse. The Tine l sign is sensitive and specific, unlike the Rolling Test, and we consider the St. Mark`s test as a good diagnostic test. In our experience, treatment with hydrodistention-corticosteroid-anesthetic infiltration achieves a rescue/improvement in a significant percentage of patients, with good medium-term (6 months) results, alleviating the pain in 39.62% of patients, which is statistically significant compared to the pre-infiltration pain.
Acknowledgements
The authors thank the Research Unit of Hospital Universitario of Burgos for providing us with as much help as we required.
References
- Amarenco G, Lanoe Y, Perrigot M, Goudal H (1987) A new canal syndrome: compression of the pudendal nerve in alcock's canal or perinal paralysis of cyclists. Presse Med 16: 399.
- Shafik A (1991) Pudendal canal syndrome: description of a new syndrome and its treatment. Report of seven cases. Coloproctology 13: 102-109.
- Rober R, Prat-Pradal D, Labat JJ, Bensignor M, Raoul S, et al. (1998) Anatomic Basis of chronic perineal pain: role of the pudendal nerve. Surg Radiol Anat 20: 93-98.
- Shafik A, Doss SH (1999) Pudendal canal: surgical anatomy and clinical implications. Am Surg 65: 176-180.
- Silbert PL, Dunne JW, Edis RH, Stewart-Wynne EG (1991) Bicycling induced pudendal nerve pressure neuropathy. Clin Exp Neurol 28: 191-196.
- Antolak SJJ, Hough DM, Pawlina W, Spinner RJ (2002) Anatomical basis of chronic pelvic pain syndrome: the ischial spine and pudendal nerve entrapment. Med Hypotheses 59: 349-353.
- Pisani R, Stubinski R, Datti R (1997) Entrapment neuropathy of the internal pudendal nerve. Report of two cases. Scand J Urol Nephrol 31: 407-410.
- Soga H, Nagata I, Murakami G, Yajima T, Takenaka A, et al. (2007) Histotopograpic study of the perineal body in elderly women: the surgical applicability of novel histological findings. Int Urogynecol J Pelvic Floor Dysfunct.
- Lien KC, Morgan DM, Delancey JO, Ashton-Miller JA (2005) Pudendal Nerve Stretch during vaginal birth: a 3D computer simulation. Am J Obstet Gyencol 192: 1669-1676.
- Benson JT, Mcclellan E (1993) The effect of vaginal dissection on the pudendal nerve. Obstet Gynecol 82: 387-389.
- Kiff ES, Barnes PR, Swash M (1984) Evidence of pudendal neuropathy in patients with perineal descent and chronic straining at stool. Gut 25: 1279-1282.
- Ho YH, Goh HS (1995) The neurophysiological significance of perineal descent. Int J Colorectal Dis 10: 107-111.
- Shafik A (1996) Pudendal canal descompression for the treatment of fecal incontinente in complete rectal prolapse. Am Surg 62: 339-343.
- Shafik A (1994) Pudendal canal descompression in the treatment of the erectile dysfunction. Arch Androl 32: 141-149.
- Shafik A (2006) Pudendal canal descompression in the treatment of the erectile dysfunction. 31 meeting of the international urogynecological association (IUGA).
- Loeser J (2003) Terapeútica del dolor. Bonica Vol. II. Ed. Mc Graw-Hill.
- Kiff E, Swash M (1984) Slowed conduction in the pudendal nerves in idiopathic (neurogénica) faecal incotinence. Br J Surg 71: 614-616.
- Beco J, Climov D, Bex M (2004) Pudendal nerve descompression in perineology: a case series. BMC Surg 30: 15.
- Galaup JP (2000) Dyschezia, pudendal neuropathy and functional pelvico-perineal disorders. Prog Urol 10: 114-122.
- González-Hidalgo M (1998) Exploración neurofisiológica del suelo de la pelvis. Rev Neurol 26: 432-438.
- Mollo M, Bautrant E, Rossi-Seignert AK, Collet S, Boyer R, et al. (2009) Evaluation of diagnostic accuracy of Colour Duplex Scanning, compared to electroneuromyography, diagnostic score and surgical outcomes, in Pudendal Neuralgia by entrapment: a prospective study on 96 patients. PAIN 142: 159-163.
- Labat JJ, Riant T, Robert R, Amarenco G, Lefaucheur JP, et al. (2008) Diagnostic Criteria for Pudendal Neuralgia by Pudendal Nerve Entrapment (Nantes Criteria). Neurourology and Urodynamics 27: 306-310.
- Nehme-Shuster H, Youssef C, Roy C, Brettes JP, Martin T, et al. (2005) Alcock´s Canal Syndrome revealing endometriosis. Lancet 366: 1238.
- Thomas E, Abiad L, Cytebal C, Gallix B, Taourel P, et al. (2002) Metastic Pudendal nerve compression presenting as atypical sciatica. J Spinal Disord Tech 15: 324.
- Wallace JM (2007) Update on pharmacotherapy guidelines for treatment of neurophatic pain. Curr Pain Headache Rep 11: 208-214.
- Max MB, Culnane M, Schafer SC (1987) Amitriptyline relieves diabetic neuropathy pain in patients with normal and depressed mood. Neurology 37: 589-596.
- Bryson HM, Wilde MI Amitriptyline (1996) A review of its pharmacological propperties and therapeutic use in chronic pain states. Drugs & Aging 8: 459-476.
- Kovacs P, Gruber H, Piegger J, Bodner G (2001) Ultrasound-Guided infiltration of the pudendal nerve: ultrasonographic technique. Dis Colon Rectum 44: 1381-1385.
- Gruber H, Kovacs P, Piegger J, Brenner E (2001) New, simple, ultrasound-guided infiltration of the pudendal nerve: topographic basics. Dis Colon Rectum 44: 1376-1380.
- Thoumas D, Leroi AM, Mauillon J, Muller JM, Benozio M, et al. (1999) Pudendal Neuralgia: ct-guided pudendal nerve block technique. Abdom Imaging 24: 488-492.
- Prendergast SA, Weiss JM (2004) Physical therapy and pudendal nerve entrapment. Advance 15: 47.
- Sánchez RAM, Arroyo A, Pérez VFA, Serrano P, Candela F, et al. (2006) Tratamiento de la proctalgia fugax con toxina botulínica: resultados en 5 pacientes. Rev Clin Esp 206: 137-140.
- Robert R, Labat JJ, Lehur PA, Glemain P, Armstrong O, et al. (1989) Clinical neurophysiologic and therapeutic remarks from anatomic data on the pudendal nerve in some cases of perineal pain. Chirurgie 115: 515-20.
- Rofael A, Peng P, Louis I, Chan V (2008) Feasibility of real-time ultrasound for pudendal nerve block in patients with chronic perineal pain. Reg Anesth Pain Med 33: 139-145.
- Mauillon J, Thoumas D, Leroi AM, Freger P, Michot F, et al. (1999) Results of pudendal nerve neurolysistransposition in twelve patients suffering from pudendal neuralgia. Dis Colon Rectum 42: 186-192.
- Shafik A (1997) Endoscopic pudendal canal decompression for the treatment of fecal incontinence due to pudendal canal syndrome. J Laparoendoscopic Adv Surg Tech A 7: 227-234.
- Valovska A, Peccora CD, Philip CN, Kaye AD, Urman RD (2014) Sacral neuromodulation as a treatment for pudendal neuralgia. Pain Physician 17: E645-E650.
- Lema R, Ricci PA (2006) Atrapamiento del nervio pudendo: un sÍndrome por conocer. Rev Chil Obstet Ginecol 71: 207-215.
- Itza Santos F, Zarza-Luciáñez D, Salinas J, Gómez Sancha F (2007) Pudendal Nerve Entrapment syndrome. Urodinamia aplicada.
- Ricci P, Lema R, Solá V, Wash A, Pardo J (2009) Infiltración del nervio pudendo guiada por tomografia axial computerizada, por vía transglútea: terapia frente al dolor ocasionado por neuralgia del nervio pudendo. Rev Chil Obstet Ginecol 74: 94-101.
- Bautrant E, De Bisschop E, Vaini-Elies V, Massonnat J, Aleman I, et al. (2003) La prise en charge moderne des néuralgies pudendales. A partir d´une série de 212 patientes et 104 interventions de décompression. J Gynecol Obstet Biol Reprod 32: 705-712.
- Speed CA (2007) Injection therapies for soft-tissue lesions. Best Pract Res Clin Rheumatol 21: 333-347.
- Mc Donald JS, Spigos DG (2008) Computed tomography-guided pudendal block for treatment of pelvic pain due to pudendal neuropathy. Obstet Gynecol 95: 306-309.
- Fannucci E, Manenti G, Ursone A, Fusco N, Mylonakou I, et al. (2009) Role of interventional radiology in pudendal neuralgia: a description of techniques and review of the literature. Radiol Med 114: 425-436.
- Dehkharghani S, vanSonnenberg E, Moore M, Hedayati P, Oyola E, et al. (2007) CTGuided Therapy of Refractory Pudendal Neuralgia: Indications, techniques and preliminare results. Am J Roentgenol 188: 114-118.
- Filler A (2008) Diagnosis and management of pudendal nerve entrapment syndromes: Impact of MR neurography and open MR-guided injections. Neurosurg Q 18: 1-6.
- Choi SS, Lee PB, Kim YC, Kim HJ, Lee SC (2006) C-arm-guided pudendal nerve block: a new technique. Int J Clin Pract 60: 553-556.
- Le Tallec de Certains H, Veillard D, Dugast J, Estébe JP, Kerdraon J, et al. (2007) Comparaison entre la latence distale motrice du nerf pudendal, la topographie de la douleur perineale et le resultat d´infiltrations. Analyse pour 53 patients. Ann Readapt Med Phys 50: 65-69.
- Popeney C, Ansell V, Renney K (2007) Pudendal entrapment as an etiology of chronic perineal pain: diagnosis and treatment. Neurourol urodyn.
- Robert R, Labat JJ, Riant T, Khalfallah M, Hamel O (2007) Neurosurgical treatment of perineal neuralgias. Adv Tech Stand Neurosur 32: 41-59.

Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences