MRI in Cesarean Scar Ectopic Pregnancy
Fatma Mohamed Awad and Ebtesam Moustafa kamal
DOI10.21767/2471-299X.1000009
1Radiology Department, Faculty of Medicine, Cairo University, Egypt
2Obstetrics and Gynecology Department, Faculty of Medicine, Zagazig University, Egypt
- *Corresponding Author:
- Fatma Mohamed Awad
Radiology Department, Faculty of Medicine, Cairo University, Egypt
Tel: 00201144180999. E-mail: ftm_wd@yahoo.co.uk
Received Date: October 15, 2015; Accepted Date: November 27, 2015; Published Date: December 05, 2015
Citation: Fatma MA,Ebtesam MK,MRI in Cesarean Scar Ectopic Pregnancy.Med Clin Rev. 2015, 1:9. doi: 10.21767/2471-299X.1000009
Copyright: © 2015 Fatma MA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
Ectopic pregnancy overall is the leading cause of death in the first trimester [1]. The incidence of pregnancy implantation in unusual sites, like cesarean scar is rising due to increased incidence of cesarean sections, pelvic inflammatory disease and dilatation and curettage [2]. Up to 72% of cesarean scar pregnancies occur in women who have had 2 or more cesarean deliveries [3-5]. The dehiscent myometrial defect may be related to incomplete healing or increased fibrosis along the uterine scar. Fibrosis occurring after multiple cesarean deliveries leads to poor vascularity, which impairs healing. Multiple cesarean deliveries also increase the risk of implantation on the scar, likely due to an increased scar surface area [4-6]. Cesarean scar ectopic pregnancy can be devastating because of complications such as uterine rupture and massive hemorrhage, leading to increased maternal morbidity and mortality [7].
Early recognition of this type of pregnancy is therefore critical in directing treatment, preventing maternal complications, and allowing successful preservation of the uterus [8]. Transvaginal Ultrasonography is the most commonly used imaging modality. Sonographic criteria, which could be helpful in diagnosis of cesarean scar pregnancy, are defined by Vial et al; however, transvaginal Ultrasonography cannot put a definitive diagnosis in all cases [9]. MRI reveals localization of the gestational sac and its relationship with adjacent organs and assesses the possibility of myometrial invasion and bladder involvement more clearly. Therefore, it provides very important information in determining the treatment plan [10].
The aim of this study is to cast the light on the role of MRI in cases of cesarean scar ectopic pregnancy as experienced in our hospital.
Materials and Methods
Patient selection
This study was conducted in the period from July, 2011, until August, 2015. It included nine patients; their ages between 37 and 45 years (mean age, 41 years). They had low-lying gestational sacs on antenatal scans and were referred to the radiology department for consultation.
Following transvaginal ultrasound, MRI was requested for confirmation of the diagnosis of cesarean scar ectopic pregnancy or for detection of placental invasion. Pelvic MRI was performed on the same or the next day of ultrasound.
The local ethics committee approved the study, and an informed written consent was obtained from each patient prior to MRI.
MRI
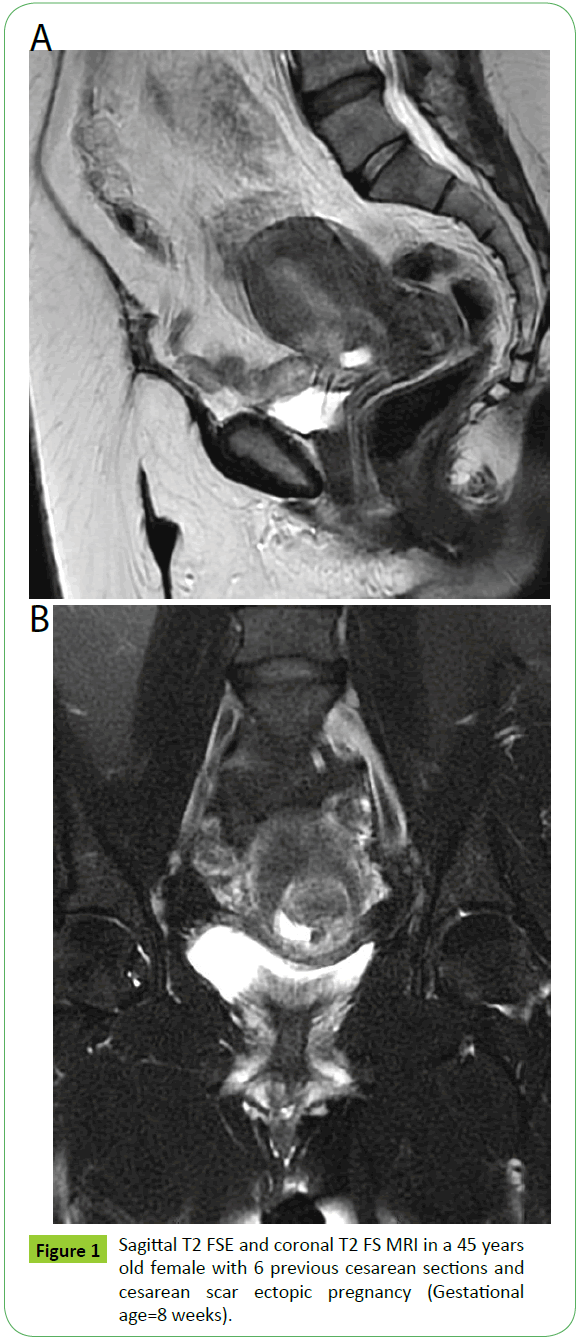
MRI studies were obtained using a 1.5 Tesla MRI (Optima MR 450W, GE Healthcare, South Carolina, USA) using a pelvic coil. The patient was asked to empty the urinary bladder prior to the study. The sequences used were sagittal T2 fast spin echo (FSE) (TR/TE, 7058/147.4 ms; slice thickness, 5 mm; matrix, 320x320) and sagittal, axial and coronal T2 FS (fat-suppressed); (TR/TE, 6500.8/147.2 ms; slice thickness, 5 mm; matrix, 320 × 320). We intended to give the patients intravenous contrast, but all refused except for one. Post contrast T1 FS images were obtained in sagittal and coronal planes (TR/TE, 453/7 ms; slice thickness, 5 mm; matrix, 300 × 300). Gadopentetate Dimeglumine (Magnevist; Schering, Berlin, Germany) was used as a contrast medium. The contrast medium was given intravenously by an automatic MRcompatible injector. The dose was 0.1 mmol/kg (Figure 1).
Interpretation of MRI
The diagnosis of Cesarean scar ectopic pregnancy was done when empty uterine and cervical cavities were noted, and a gestational sac seen embedded within the site of cesarean scar, with thin myometrium adjacent to the sac. Bulging of the sac through the myometrium, with or without invasion of the urinary bladder, was checked in all cases. No bladder invasion was detected in any of the patients.
Management
Conservative management; systemic methotrexate (1 mg/ kg) was the first line of treatment for the patients, followed by evacuation of retained products of conception (ERPOC) in seven patients. Follow up β-HCG testing was performed after six to eight weeks, reverting to normal levels. In the other two patients, hysterectomy was done due to failed methotrexate therapy.
Results
This study included nine patients with Cesarean scar ectopic pregnancy. On transvaginal ultrasound, cesarean scar ectopic pregnancy was diagnosed in six cases, however due to remarkable thinning of the myometrium adjacent to the gestational sacs (<5 mm), MRI was requested to exclude placental invasion. In the other three patients, no myometrial thinning was noted on ultrasound; however, MRI was requested to confirm the diagnosis; as incomplete abortion couldn’t be totally excluded on sonographic basis. MRI confirmed the diagnosis of cesarean scar ectopic pregnancy in all the patients with no evidence of placental invasion to the uterine wall or urinary bladder.
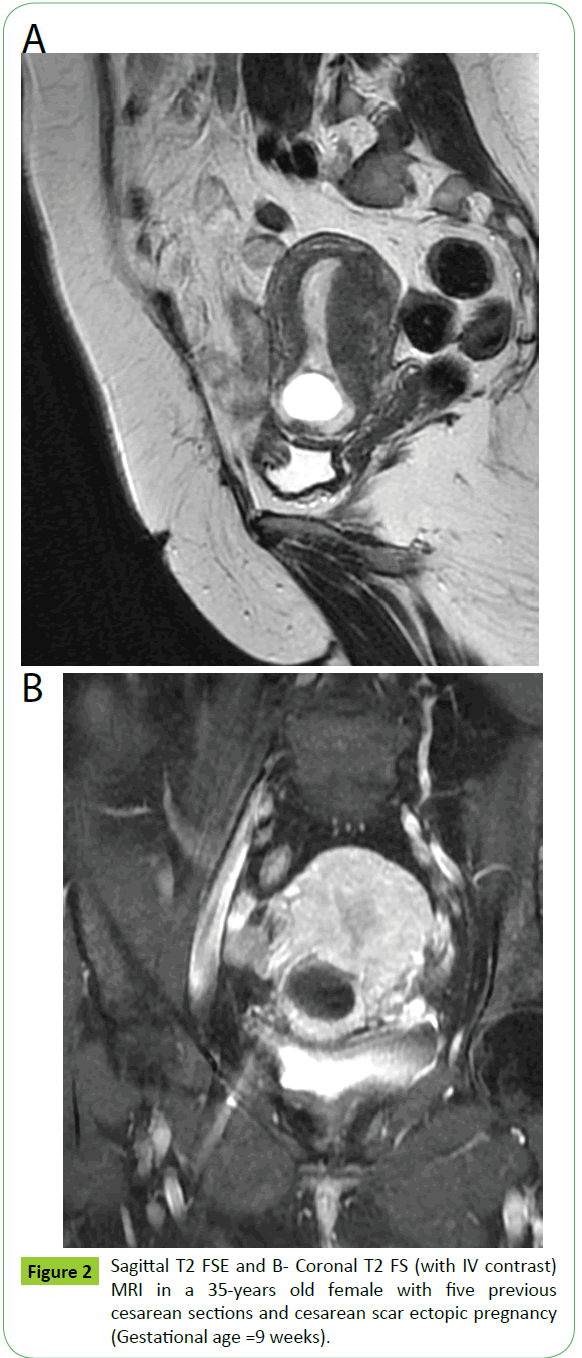
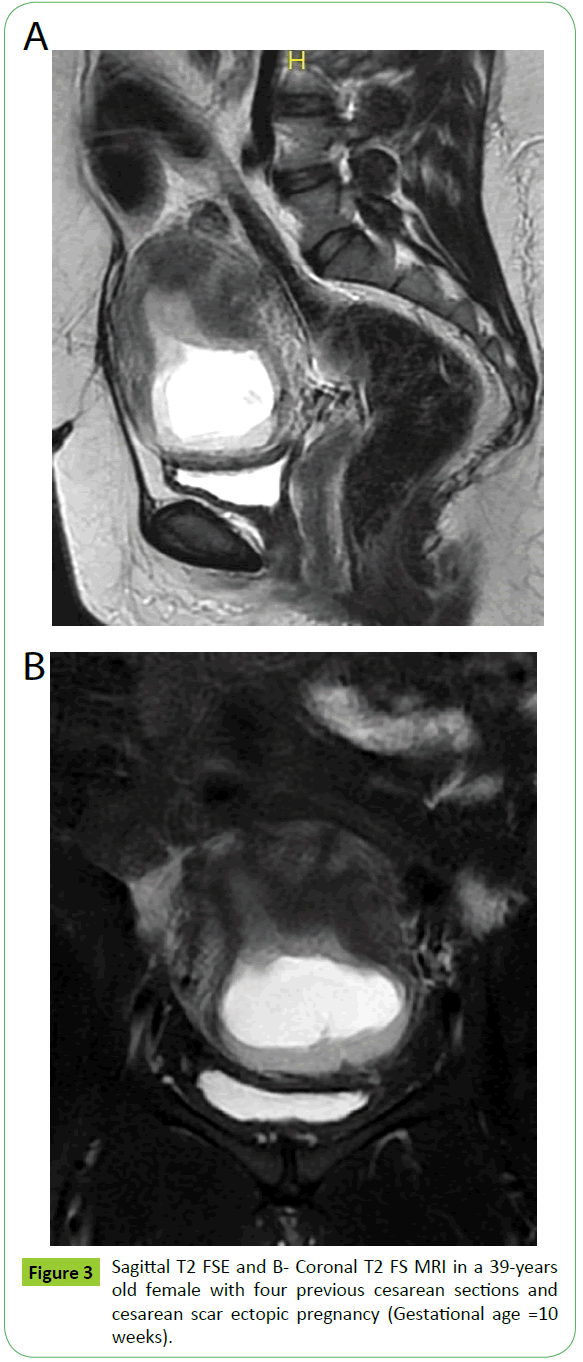
The gestational age of the patients ranged from 7–10 weeks. The number of previous cesarean sections ranged from four to seven sections (Figures 2 and 3).
The clinical presentation of the patients is shown in Tables 1,2 and 3 illustrate the sonographic and MRI findings in the cases.
| Clinical presentation | Number of patients (percentage) |
|---|---|
| Low lying gestational sac on antenatal ultrasound | 9 |
| Abdominal Pain | 1 |
| Vaginal Bleeding | 9 |
Table 1 Clinical presentation of the patients in the study: *One of the patients complained of vaginal bleeding with dull-aching lower abdominal pain.
| Findings | Number of Patients |
|---|---|
| Empty uterus with a clearly visualized endometrium. | 9 |
| Empty cervical canal. | 9 |
| Gestational sac at the presumed site of the cesarean scar. | 9 |
| Thinned myometrium between the gestational sac and the urinary bladder. | 6 (<5mm) |
| Cardiac Pulsations in the gestational sac | None |
Table 2 Sonographic findings in the patients.
| Findings | Number of Patients |
|---|---|
| Empty uterine and cervical cavities with a gestational sac seen embedded at the site of cesarean scar), with thin myometrium adjacent to the sac. | 9 |
| Bulging of the sac through the myometrium. | None |
| Disrupted bladder wall integrity. | None |
Table 3 MRI findings in the patients.
Discussion
Cesarean scar ectopic pregnancy is considered among the rarest forms of ectopic pregnancies. Its prevalence is estimated between 1 per 1800 and 1 per 2226 pregnancies [3,11].
Up to 72% of cesarean scar pregnancies occur in women who have had 2 or more caesarean deliveries [3-5]. All the patients included in our study (100%) had at least four previous cesarean sections, the maximum number being seven.
Women with cesarean scar pregnancy may present with vaginal bleeding. Abdominal pain may not always be present [12]. One out of nine patients (11%) in this study presented with abdominal pain, while the rest (89%) presented with vaginal bleeding in the first trimester. On antenatal scan, all the patients (100%) had lowlying gestational sacs.
Transvaginal ultrasound has a reported sensitivity of 84.6% [12] and remains the imaging modality of choice for diagnosis of cesarean scar pregnancy, though MRI may play a greater role in its evaluation. The superior soft tissue characterization and anatomical information provided by MRI allows patients and clinicians to consider conservative management as initial therapy, especially with the increasing availability of minimally invasive uterine artery embolization [13]. MRI can accurately detect the exact location of pregnancy, thus confirming the diagnosis [1,14]. Differential diagnosis of caesarean scar ectopic pregnancy includes cervical pregnancy, early placenta accreta and incomplete abortion, when patients may mistakenly undergo curettage leading to life threatening hemorrhage [2]. In our study, MRI was done for six patients (67%) with thinned out myometrium adjacent to the gestational sac to check for placenta accreta. In the other three patients (33%), it was done to confirm the diagnosis; as incomplete abortion couldn’t be totally excluded on sonographic basis.
Wu et al in their report of a case of cesarean scar pregnancy followed almost the same steps as in this study. The findings on initial ultrasound examination raised the suspicion of a cesarean scar ectopic pregnancy with possible isthmic-cervical involvement, while the proximity of the sac to the urinary bladder raised the possibility of urinary bladder involvement; however ultrasound examination could not exclude this possibility with certainty. Further evaluation with MRI was performed, followed by medical management with surgical intervention.
We wanted to do post contrast MRI for all the patients to aid in the diagnosis of cesarean scar ectopic pregnancy, however, all refused except for one patient. The most recent study regarding use of intravenous contrast in MRI done for patients with cesarean scar pregnancy was that by Huang et al [15], who concluded that contrast-enhanced MRI could be used as a reliable adjunct and initial imaging modality for diagnosing CSP in selected cases. The imaging features of contrast-enhanced MRI may result in a more accurate diagnosis before specific treatment for CSP
Conclusion
Transvaginal ultrasound is the first step for diagnosis of cesarean scar ectopic pregnancy. However, MRI should be kept in mind if the diagnosis cannot be made with certainty or if placenta accreta cannot be excluded on sonographic basis.
References
- Koroglu M, Kayhan A, Soylu FN, Erol B, Schmid-Tannwald C, et al. (2013) MR imaging of ectopic pregnancy with an emphasis on unusual implantation sites. Jpn J Radiol 31: 75-80.
- Timor-Tritsch IE, Monteagudo A (2012) Unforeseen consequences of the increasing rate of cesarean deliveries: early placenta accreta and cesarean scar pregnancy. A review. Am J Obstet Gynecol 207: 14-29.
- Ash A, Smith A, Maxwell D (2007) Caesarean scar pregnancy. BJOG 114: 253-263.
- Jurkovic D, Hillaby K, Woelfer B, Lawrence A, Salim R, et al. (2003) First trimester diagnosis and management of pregnancies implanted into the lower uterine segment Cesarean section scar. Ultrasound Obstet Gynecol 21: 220-227.
- Maymon R, Halperin R, Mendlovic S, Schneider D, Herman A (2004) Ectopic pregnancies in a Caesarean scar: review of the medical approach to an iatrogenic complication. Hum Reprod Update 10: 515-523.
- Rosen T (2008) Placenta accreta and cesarean scar pregnancy: overlooked costs of the rising cesarean section rate. Clin Perinatol 35: 519-529.
- Daniel A Osborn, Todd R Williams, Brian M Craig (2012) Cesarean Scar Pregnancy: Sonographic and Magnetic Resonance Imaging Findings, Complications, and Treatment. J Ultrasound Med 31: 1449-1456.
- Fylstra DL (2002) Ectopic pregnancy within a cesarean scar: a review. Obstet Gynecol Surv 57: 537-543.
- Vial Y, Petignat P, Hohlfeld P (2000) Pregnancy in a cesarean scar. Ultrasound Obstet Gynecol. 16: 592-593.
- Bozkurt DK, Bozkurt M, Sahin L (2014) Magnetic Resonance Imaging in Addition to Ultrasound in the Diagnosis of Cesarean Scar Ectopic Pregnancy. J Med Cases 5: 329-333.
- Seow KM, Huang LW, Lin YH, Lin MY, Tsai YL et al. (2004) Cesarean scar pregnancy: Issues in management. Ultrasound Obstet Gynecol23: 247-253.
- Rotas MA, Haberman S, Levgur M (2006) Cesarean scar ectopic pregnancies: Etiology, diagnosis and management. Obstet Gynecol 107: 1373-1381.
- Wu R, Klein MA, Mahboob S, Gupta M, Katz DS (2013) Magnetic Resonance Imaging as an Adjunct to Ultrasound in Evaluating Cesarean Scar Ectopic Pregnancy. J Clin Imaging Sci 3: 16.
- Osborn DA, Williams TR, Craig BM (2012) Cesarean scar pregnancy: Sonographic and magnetic resonance imaging findings, complications, and treatment. J Ultrasound Med 31: 1449-1456.
- Huang Q, Zhang M, Zhai RY (2014) The use of contrast-enhanced magnetic resonance imaging to diagnose cesarean scar pregnancies. Int J Gynaecol Obstet 127: 144-146.

Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences